We created the da Vinci 5 platform with the goal of helping surgeons provide the best minimally invasive surgery to as many patients as possible. Redesigned from the inside out, da Vinci 5 brings more than 150 design innovations and 10,000x the computing power1 to deliver greater surgeon autonomy,2 more streamlined OR workflow, and advanced data analytics. With an eye toward improving patient outcomes now and into the future, we have enabled real-time collaboration and state‑of‑the‑art analytic tools that deliver objective insights to personalize surgeon learning.
Designed for better outcomes by enhancing surgical senses
Better patient outcomes: this goal underscores everything we do. We aim to deliver these outcomes through enhanced surgical senses—by enabling Force Feedback technology, the best da Vinci vision system, and improved ergonomics.<sup>3</sup>
<div id="forcefeedback"> Force Feedback: first-of-its-kind technology</div>
Surgeons with varying experience levels can deliver up to 43% less force on tissue<sup>4</sup> by sensing the push and pull at the instrument tip through Force Feedback technology. The instrument sensors are able to generate post-operative, objective performance measurements on tissue for personal and training insights. Surgeons who were early in their learning curve made fewer errors, performed faster suturing, and with less tissue trauma when using instruments with Force Feedback technology during simulated suturing tasks.<sup>5</sup>The best da Vinci vision system
The da Vinci 5 vision system delivers our best, most realistic 3D image—with better color and resolution.<sup>3</sup>Our most ergonomic system ever
An immersive, reengineered console with a greater range of motion and wider range of ergonomic settings<sup>3</sup> designed to offer more comfort<sup>8</sup> for the surgeon.More surgeon autonomy<sup>2</sup> and streamlined workflow
Over 150 design innovations may lead to reduced procedure times<sup>9</sup> with potential to increase throughput. Greater surgeon autonomy<sup>2</sup> and streamlined workflow can help hospitals address staffing challenges. The result: more operational efficiency – critical in helping you scale da Vinci surgery across the patients you serve.Greater surgeon autonomy<sup>2</sup>
Through component integration and control at their fingertips, surgeons can manage all components needed for surgery from their console, helping to reduce care team workload.<sup>10</sup>Streamlined workflows
<p>Da Vinci 5 is easy to learn, with simplified setup, guided tool change, and task automation. A universal user interface extends across all three system components to enhance care team experience. </p>Reduced procedure time<sup>9</sup>
<p>Da Vinci 5 refinements benefit new users—as well as expert users. Over 150 design innovations may result in increased throughput and the potential to add da Vinci surgeries beyond traditional OR timetables.</p>Objective insights, personalized action—enabled by My Intuitive+
Objective insights, personalized action—enabled by My Intuitive+
Digital tools currently available through My Intuitive+ allow surgeons to collaborate in real time and use surgical video and data collected to objectively understand surgical performance and receive personalized training.
Telepresence: bridge the gap between distance and connection
Enable real-time case observation, collaboration, and mentoring with Telepresence. Surgeons can schedule a session prior to a case for enhanced learning, as well as stay up-to-date with the latest surgical approaches across the da Vinci community.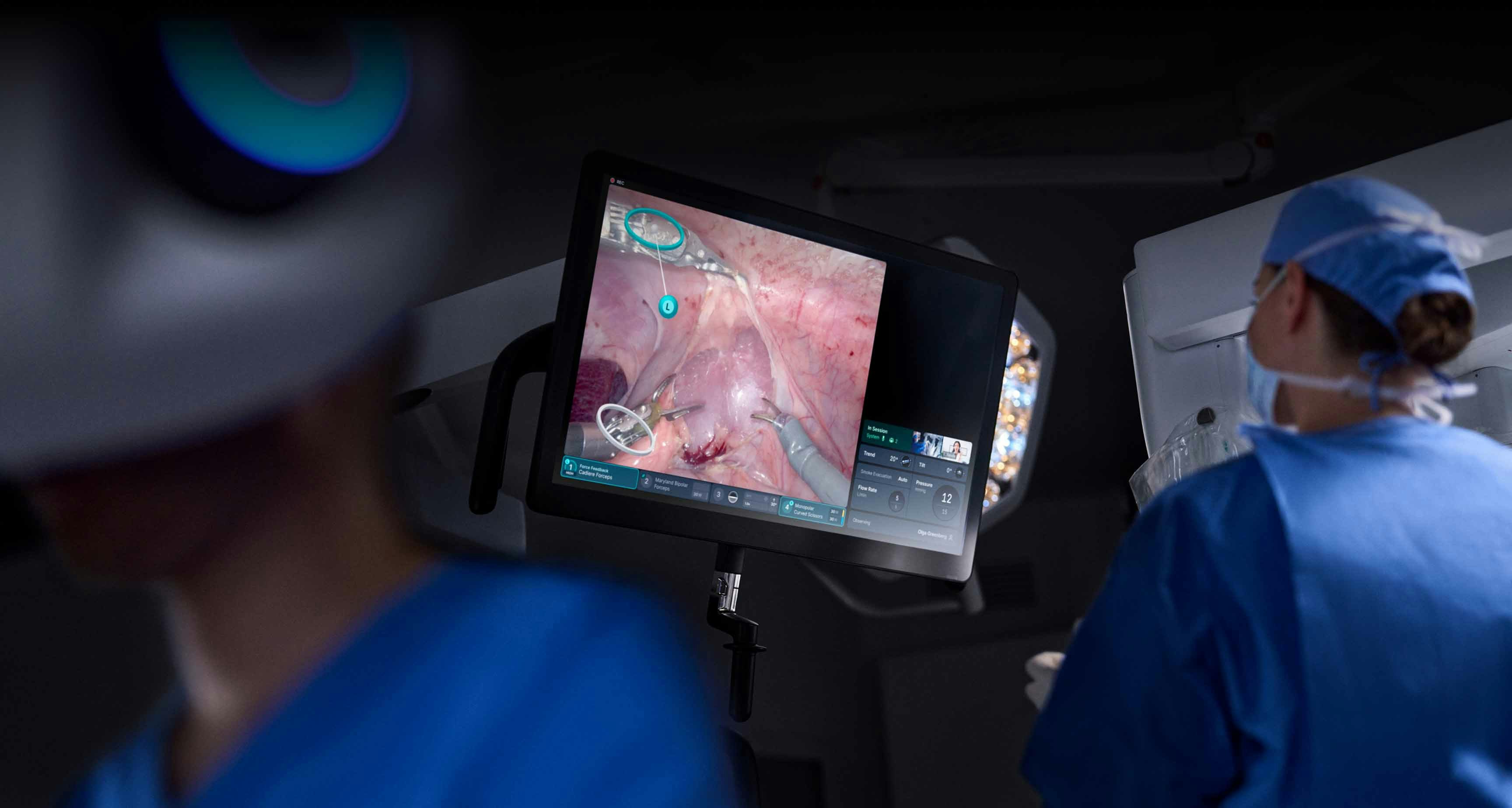
Case Insights: understand surgical performance like never before
Know more about personal and program performance through da Vinci 5’s rich data set and objective skills assessment. Case insights uses AI to evaluate da Vinci system data, kinematic movement, and video—creating objective insights to help surgeons elevate their skills, with the goal of improving patient outcomes.SimNow 2: build proficiency in a consequence-free environment
Learn and maintain skills through over 65 simulation exercises; the self-guided curriculum helps surgeons shorten their learning curve and increase confidence while gaining expert proficiency—with better skill acquisition and training outcomes.<sup>13</sup>
Smarter together:
Intelligently integrated hardware
Smarter together:
Intelligently integrated hardware
- Compared to da Xi. Data on file at Intuitive.
- Based on a preclinical human factors study comparing surgeons and care team use of da Vinci 5 to da Vinci Xi. Data on file at Intuitive.
- Compared to da Vinci X, Xi, and SP systems. Data on file at Intuitive.
- Based on preclinical data comparing High to Off settings for retraction. Awad MM, Raynor MC, Padmanabhan-Kabana M, Schumacher LY, Blatnik JA. Evaluation of forces applied to tissues during robotic-assisted surgical tasks using a novel Force Feedback technology. Surg Endosc. 2024;38(10):6193-6202. doi:10.1007/s00464-024-11131-z.
- Based on pre-clinical data comparing new surgeons with and without instruments with Force Feedback technology during simulated suturing tasks. Servais EL, Rashidi L, Porwal P, Garibaldi M, Hung AJ. Novel Force Feedback technology improves suturing in robotic-assisted surgery: a pre-clinical study. Surg Endosc. 2025;39(2):1217-1226. doi:10.1007/s00464-024-11472-9.
- Average forces reported are within this sensing range.
- Contraindication: use of the Force Feedback needle driver is contraindicated in hysterectomy and myomectomy due to the risk of vaginal bleeding requiring hospital readmission and/or the need for additional procedures. The use of non-force feedback needle drivers is recommended for suturing in these procedures.
- Based on a preclinical human factors study comparing 13 surgeons’ use of da Vinci 5 to da Vinci Xi. Data on file at Intuitive.
- Compared to da Vinci Xi. The result presented is an aggregate, qualitative analysis of data from 53 first-in-human-use cases on da Vinci 5, which included 23 surgeons at novice, intermediate, and expert experience levels. Results may vary by procedure type and surgeon experience level. Data on file at Intuitive.
- Based on a preclinical human factors study comparing 11 surgeons and 22 care team members’ use of da Vinci 5 to da Vinci Xi. Data on file at Intuitive.
- Based on a preclinical human factors study comparing 8 care team members’ use of da Vinci 5 to da Vinci Xi. Data on file at Intuitive.
- SAGES 2024 Cleveland "Remote Telepresence Verses In-Person Case Observations for Robotic Surgery Training Pathways" Andrea Pakula, MD, MPH, FACS, Travis Trail, Gretchen Purcell Jackson MD, PhD.
- Using the GEARS Scale (Global Evaluative Assessment of Robotic Skills). Raison N, Harrison P, Abe T, Aydin A, Ahmed K, Dasgupta P. Procedural virtual reality simulation training for robotic surgery: a randomised controlled trial. Surg Endosc. 2021;35(12):6897-6902. doi:10.1007/s00464-020-08197-w.





















